Opioid replacement therapy: definition, drugs, and effectivity
Table of content
- What is opioid replacement therapy (opioid substitution treatment)?
- How does opioid replacement therapy work?
- What are the drugs used in opioid replacement therapy?
- How is opioid replacement therapy done?
- Is opioid replacement therapy effective?
- What is the difference between opioid replacement therapy and medication-assisted treatment?
- What is the difference between opioid replacement therapy and opioid agonist therapy?

Opioid replacement therapy, or opioid substitution treatment, is a long-term treatment for opioid dependence that involves administering long-acting opioid agonist medications under medical supervision. It provides sustained stabilization, reducing cravings and withdrawal while enabling individuals to focus on recovery, social reintegration, and overall well-being.
Drugs used in opioid replacement therapy include methadone, buprenorphine, and naltrexone. These medications help manage opioid dependence and minimize the harm associated with illicit opioid use.
Effectivity of opioid replacement therapy lies in its ability to reduce withdrawal symptoms, cravings, and illicit opioid use while preventing overdose risks. By stabilizing individuals with long-acting medications such as methadone and buprenorphine, opioid replacement therapy supports long-term recovery and improves treatment retention.
What is opioid replacement therapy (opioid substitution treatment)?
Opioid replacement therapy is a harm reduction approach designed to help individuals struggling with opioid addiction. Another term used for it is opioid substitution treatment. Since the late 1960s, this therapy has been extensively researched and implemented in many countries to replace the harmful and illicit use of opioids. The therapy involves the substitution of opioids with long-acting medicines under medical supervision.
The research article “Psychosocial interventions in opiate substitution treatment services: does the evidence provide a case for optimism or nihilism?” authored by Ed Day and Luke Mitcheson, published in the journal Addiction in 2016, highlights that combining opioid replacement therapy with psychosocial interventions enhances treatment effectiveness. The key is to customize these therapies to fit each person’s specific needs throughout their opioid addiction treatment.
What is the goal of opioid replacement therapy treatment?

The goal of opioid replacement therapy treatment is to stabilize patients with opioid dependence, reduce drug withdrawal symptoms and cravings, minimize the risk of overdose, and decrease harmful behaviors associated with drug use. In addition, opioid replacement therapy aims to improve patients’ overall physical and psychological health, support social functioning, and facilitate their reintegration into society, even if achieving complete abstinence is not immediately feasible.
Is opioid replacement therapy substituting one addiction for another?
No, opioid replacement therapy is not substituting one addiction for another. While opioid replacement therapy (ORT) involves the use of opioid-based medications such as methadone or buprenorphine, these treatments are administered in controlled, medically supervised settings to stabilize individuals, reduce cravings, and prevent withdrawal symptoms.
Methadone addiction is a concern often raised in discussions about ORT. However, when methadone is used as prescribed in a structured treatment program, it functions as a harm reduction tool rather than a source of compulsive drug use.
The article “Harm Reduction,” published by the Substance Abuse and Mental Health Services Administration (SAMHSA) and last updated on October 29, 2024, recognizes ORT as an effective harm reduction strategy. Additionally, the agency emphasizes the importance of distributing opioid overdose reversal medications to individuals at risk or those in a position to respond to an overdose, further strengthening public health efforts to combat the opioid crisis.
Opioid replacement therapy is a legitimate and effective medical treatment for opioid addiction. According to the article “Substance Use Disorder Treatment Options,” published by the Substance Abuse and Mental Health Services Administration (SAMHSA) last updated on April 11, 2024, medications used in ORT are evidence-based treatments that go beyond merely replacing one drug with another. It’s about providing a safer, more stable alternative to illicit opioids, reducing harm, and supporting long-term recovery.
How does opioid replacement therapy work?
Opioid replacement therapy works by using long-acting opioid medications to stabilize brain receptors, prevent withdrawal symptoms, and reduce cravings without inducing the intense euphoria associated with illicit opioid use. Unlike short-acting opioids such as heroin or fentanyl, which cause rapid highs followed by severe withdrawal, opioid replacement therapy (ORT) medications maintain steady opioid activation, allowing individuals to function normally and reducing compulsive drug-seeking behavior.
According to the WHO/UNODC/UNAIDS position paper “Substitution maintenance therapy in the management of opioid dependence and HIV/AIDS prevention” published by the World Health Organization, United Nations Office on Drugs and Crime, Joint United Nations Programme on HIV/AIDS, 2004, ORT operates through pharmacological substitution, where prescribed opioid agonists like methadone (a full agonist) and buprenorphine (a partial agonist) activate opioid receptors in a controlled and sustained manner. This prevents withdrawal and minimizes cravings while blocking the euphoric effects of other opioids, discouraging misuse. Additionally, naltrexone, an opioid antagonist, is sometimes used in ORT for individuals who have detoxed and seek complete abstinence, as it blocks opioid effects entirely.
Beyond its pharmacological effects, ORT plays a critical role in harm reduction. By replacing illicit opioids with safer, medically supervised alternatives, ORT helps prevent overdose, injection-related diseases (HIV, hepatitis C), and criminal activities linked to opioid dependence. ORT medications are administered orally or sublingually, eliminating the dangers of unsafe injection practices. Furthermore, they provide a predictable and controlled opioid effect, reducing fluctuations that lead to addiction cycles.
What are the risks of opioid replacement therapy?

The risks of opioid replacement therapy are listed below.
- Accessibility: Access to opioid replacement therapy (ORT) is limited, especially in rural or underserved areas. Treatment facilities are scarce, and patients have to travel long distances to access medications, discouraging consistent participation in the treatment.
- Side effects: Long-term use of ORT medications results in side effects like constipation, weight gain, and potential cardiovascular issues. Buprenorphine, while safer in terms of overdose risk, causes headaches, dizziness, or nausea.
- Psychosocial risks: While ORT stabilizes patients physically, it doesn’t directly address the psychological aspects of addiction. Without proper psychosocial support and therapy, patients do not fully recover, and the risk of relapse remains high.
- Stigma and discrimination: People seeking ORT often face stigma due to their opioid use disorder, which prevents them from accessing services. This stigma is prevalent in healthcare settings, where people are not fully supported or understood by healthcare providers.
- Overdose risk: Overdose risk is a significant concern for patients undergoing opioid substitution therapy (OST). These individuals face a higher risk of overdose, particularly during the initiation of OST and after cessation of treatment. The risk of fatal overdose is notably higher for opioid-dependent patients, especially for those with comorbidities that complicate their treatment or response to the medication. These patients are more vulnerable to both non-fatal and fatal overdoses due to the combination of their opioid dependence and underlying health conditions.
- Adverse drug-drug interactions (DDIs): DDIs present significant risks for patients on OST. Interactions with medications such as antidepressants, anxiolytics, and antiarrhythmics increase the risk of cardiovascular issues, including ventricular arrhythmias and cardiac failure. Additionally, combining OST medications with other drugs, like anxiolytics, heightens the risk of respiratory depression, which leads to respiratory failure.
- Misuse of opioid substitution therapy (OST) medication: Misuse of OST refers to the inappropriate use of drugs like methadone or buprenorphine that are intended to help individuals recover from opioid addiction. Misuse involves behaviors such as taking higher doses than prescribed, using the medication without a prescription, or diverting the medicines to others for non-medical purposes. Additionally, it leads to increased crime and the spread of blood-borne viruses, exacerbating public health challenges, as evident in the research article “Addressing misuse and diversion of opioid substitution medication: guidance based on systematic evidence review and real-world experience” authored by Nat Wright et al. published in the journal Journal of Public Health in 2016.
How long does opioid replacement therapy last?
Opioid replacement therapy lasts variably for different individuals, based on their needs and clinical guidelines. The article “Principles of Drug Addiction Treatment: A Research-Based Guide (Third Edition)” published by the National Institute on Drug Abuse (NIDA), last revised January 2018, recommends a minimum of 12 months for optimal effectiveness, as premature discontinuation increases the risk of relapse and overdose.
According to the study “Opioid treatment of opioid addiction” authored by Michael McDonough, published in the journal Australian Prescriber in 2013, patients who remain in opioid replacement therapy (ORT) for at least 12 months have higher success rates, in comparison to those who discontinue prematurely.
What are the drugs used in opioid replacement therapy?
The drugs used in opioid replacement therapy are listed below.
- Methadone
- Buprenorphine
- Naltrexone
Methadone
Methadone is a Food and Drug Administration (FDA) approved medication used for both severe pain management and opioid use disorders (OUD) treatment. Methadone is a long-acting schedule II controlled substance, strictly regulated due to its opioid composition. When administered properly, methadone is a safe and effective medication that supports recovery and enables individuals to live fulfilling lives. According to the article “Methadone” published by the Substance Abuse and Mental Health Services Administration (SAMHSA), last updated on 29 March 2024, its effectiveness is maximized when integrated into a comprehensive treatment plan that includes counseling and behavioral therapies.
Methadone is a frequently used medication in opioid replacement therapy (ORT), available in liquid, tablet, and injectable forms, though oral administration is most common. The syrup formulation is particularly useful for supervised dosing because liquids are more difficult to conceal than tablets, as those are placed under the tongue. A 2013 article, “Opioid treatment of opioid addiction” authored by Michael McDonough, published in the journal Australian Prescriber, notes that oral methadone has approximately 70% bioavailability. The study further recommends a 30% dose reduction for patients who cannot take anything orally.
The chapter “Methadone Maintenance Treatment” in the book Clinical Guidelines for Withdrawal Management and Treatment of Drug Dependence in Closed Settings, published by the World Health Organization in 2009, outlines the structured approach to methadone dosing in opioid replacement therapy. The initial dose is kept low and gradually increased to reach a maintenance dose, which effectively prevents withdrawal symptoms without inducing euphoria. Typically, the first dose ranges from 10-30 mg, with higher doses administered to those with recent opioid use. To minimize overdose risks, particularly in the critical first two weeks, daily doses do not exceed 40-60 mg.
Patients require close monitoring for three to four hours after the first dose to detect signs of overdose or withdrawal. If necessary, the dose increases by 5-10 mg every three days, ensuring that the weekly increment does not exceed 20 mg. Achieving the optimal maintenance dose—generally between 60-120 mg— takes several weeks and is tailored to each patient’s history of opioid use. This gradual adjustment ensures a safe and effective transition to methadone maintenance therapy, reducing the risk of relapse while supporting long-term recovery.
Buprenorphine
Buprenorphine, a Food and Drug Administration (FDA) approved schedule III synthetic opioid drug developed in the late 1960s, is primarily used for pain management and opioid use disorder (OUD) treatment. It’s the first OUD medication that is prescribed or dispensed in standard physician offices, greatly expanding treatment access.
Buprenorphine plays a key role in opioid replacement therapy, replacing more potent opioids like heroin with a safer option. It is a partial opioid agonist, meaning it produces limited effects compared to full agonists. While it provides pain relief, it is less likely to cause euphoria. It is commonly used in ORT to help manage opioid dependence. Its ceiling effect limits its potential for overdose, making it a safer treatment option. However, its use in opioid dependence treatment is highly regulated and closely monitored to ensure safety and prevent misuse.
Buprenorphine is available in various formulations, including sublingual tablets, buccal films, transdermal patches, and injectable forms. Since oral absorption is poor, buprenorphine is typically administered sublingually.
Buprenorphine treatment begins with a low initial dose, typically between 2 mg and 4 mg, with gradual weekly titration to ensure patient safety and effectiveness, as evidenced in the article “Buprenorphine” by Rachna Kumar et al. published in the book StatPearls, last updated June 8, 2024. If well-tolerated, doses are then increased in 2–4 mg increments until 24-hour stabilization is achieved. The maintenance phase involves adjusting the dose based on individual needs, usually stabilizing between 8–12 mg daily, though it does not exceed 24 mg per day due to the risk of diversion. The article further suggests that longer-term buprenorphine treatment over 90 days leads to better recovery outcomes.
The research article “Abuse liability of intravenous buprenorphine/naloxone and buprenorphine alone in buprenorphine-maintained intravenous heroin abusers” authored by Sandra D Comer published in the journal Addiction in 2010, highlights that naloxone is commonly combined with buprenorphine in sublingual formulations like Suboxone (buprenorphine/naloxone in a 4:1 ratio) to prevent intravenous misuse of buprenorphine. When taken sublingually as prescribed, naloxone has minimal absorption and does not interfere with buprenorphine’s therapeutic effects.
Buprenorphine is used in opioid substitution therapy (OST) for pregnant women, where it is associated with a lower incidence of neonatal withdrawal syndrome compared to other opioid treatments.
Naltrexone
Naltrexone is an opioid blocker approved by the Food and Drug Administration (FDA) for managing both opioid dependence and alcohol use disorder. Developed in 1963 and patented in 1967, naltrexone received US approval in 1984. According to the research article “Acute Pain Management for People with Opioid Use Disorder” by Kathleen Broglio and Marianne Matzo published in the American Journal of Nursing in 2018, nearly 10,000 patients in the US received naltrexone in 2016 as part of acute pain management during opioid addiction treatment.
Naltrexone blocks opioid effects, thereby preventing intoxication and the development of physiological dependence in users. This is evidenced in the research article “Naltrexone” authored by Dharminder Singh and Abdolreza Saadabadi and published in the book StatPearls in 2023. The medication is available in tablet form and depot injections. Naltrexone is administered orally regardless of food intake, and monthly intramuscular injections. The authors advise against naltrexone’s administration via subcutaneous, intravenous, or fatty tissue injections.
According to the article “Naltrexone” published by the Substance Abuse and Mental Health Administration (SAMHSA) last updated on March 29th, 2024, to minimize the risk of withdrawal symptoms associated with opioid use disorder (OUD), patients need to wait at least 7 days after their last use of short-acting opioids or 10 to 14 days after long-acting opioids before beginning naltrexone.
The treatment with naltrexone begins with an initial lower dose of 25 mg once daily for a few days, then is increased to 50 mg once daily, 100 mg every other day, 150 mg every 3rd day, or 380 mg depot injection for monthly administration.
How is opioid replacement therapy done?
Opioid replacement therapy is done by administering long-acting opioid medications to replace short-acting and illicit opioids. The process begins with a patient assessment, where medical history, substance use patterns, and diagnostic tests confirm opioid dependence. At effective doses, these medications prevent other opioids like heroin from producing a high typically known as the blockade effect.
If deemed suitable, the patient enters the induction phase, during which a low dose of methadone, naloxone and buprenorphine, or naltrexone alone is administered under medical supervision. The dose is gradually increased over several days to relieve withdrawal symptoms while minimizing the risk of sedation or overdose.
Once stabilized, the patient moves into the maintenance phase, where the dose is adjusted until cravings and withdrawal symptoms are controlled. During this phase, which lasts for months or years, regular medical supervision, urine drug testing, and counseling support are provided to ensure adherence and prevent misuse. If a dose is effective and has no side effects, it need not be reduced unless planned. Reducing it makes it less effective. However, if someone chooses to taper off slowly, they are supported in their decision.
Who supervises opioid replacement therapy?
Opioid replacement therapy is supervised differently across countries due to variations in healthcare systems, policies, regulations, and cultural attitudes toward addiction treatment. In the United States, opioid treatment programs (OTPs) need to be certified by the Substance Abuse and Mental Health Services Administration (SAMHSA) and are often supervised by addiction specialists, and primary care physicians.
In addition, OTPs need to be registered with the Drug Enforcement Administration (DEA) to dispense controlled substances such as methadone or buprenorphine. Finally, state-level supervision is provided by state opioid treatment authorities, which license OTPs and enforce local regulatory compliance. Together, these entities form a multi-tiered regulatory framework ensuring safety, quality, and accountability in opioid treatment services.
Is opioid replacement therapy effective?
Yes, opioid replacement therapy is effective. A 2020 study titled “Comparative Effectiveness of Different Treatment Pathways for Opioid Use Disorder” by Sarah E. Wakeman et al. published in the journal JAMA Network Open, demonstrated that opioid replacement therapy with medications such as buprenorphine and methadone significantly reduces the risk of overdose and serious opioid-related acute care use. The study found a substantial reduction in overdose risk for patients on these medications: 76% at three months and 59% at twelve months compared to those receiving no medication.
Another research article, “The effectiveness of opioid substitution treatments for patients with opioid dependence: a systematic review and multiple treatment comparison protocol” by Brittany Burns Dennis et al. published in the journal Systematic Reviews in 2014, highlights that methadone maintenance therapy (MMT) is the most commonly used opioid substitution therapy, with reported effectiveness varying between 20% and 70% across studies.
What is the difference between opioid replacement therapy and medication-assisted treatment?
The difference between opioid replacement therapy and medication-assisted treatment lies in their scope, treatment goals, and medication use. Opioid replacement therapy (ORT) is a subset of medication-assisted treatment (MAT) that specifically treats opioid use disorder (OUD) by replacing illicit opioids with safer, long-acting alternatives such as methadone or buprenorphine to reduce withdrawal symptoms, minimize illicit drug use, and lower overdose risk. In contrast, MAT is a comprehensive treatment approach for substance use disorders (SUDs) that integrates Food and Drug Administration (FDA) approved medications with behavioral therapy and counseling. It is particularly effective for managing opioid use disorder (OUD), alcohol use disorders (AUD), and nicotine dependence, addressing both the physiological and psychological aspects of addiction.
ORT focuses on harm reduction and stabilization, often as a long-term maintenance therapy where certain individuals remain on medication indefinitely, though gradual tapering is an option. In contrast, medication assisted treatment (MAT) is more recovery-oriented, aiming for long-term sobriety by integrating psychosocial support, behavioral interventions, and relapse prevention strategies to help individuals regain control of their lives.
In ORT, patients do not participate in broader recovery programs, as their treatment primarily focuses on opioid maintenance, sometimes extending for years. In contrast, MAT is typically integrated into a comprehensive rehabilitation program that includes behavioral counseling, therapy, and other supportive services to promote lasting recovery.
ORT utilizes medications such as methadone, buprenorphine, and naltrexone. In contrast, MAT is primarily employed in treating addictions to opioids, nicotine, and alcohol. It includes ORT drugs (methadone, buprenorphine, and naltrexone) along with acamprosate, disulfiram, bupropion, and varenicline.
What is the difference between opioid replacement therapy and opioid agonist therapy?
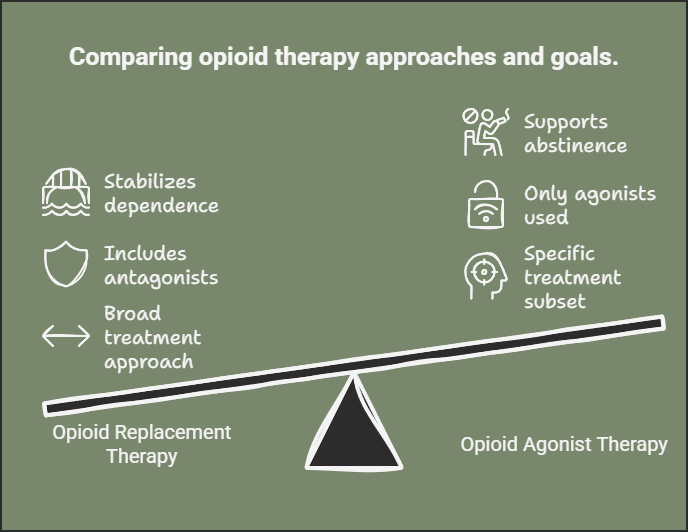
The difference between opioid replacement therapy and opioid agonist therapy lies in their scope, treatment goals, and the types of medications used. Opioid replacement therapy (ORT) is a broad treatment approach that involves substituting harmful opioids like heroin with safer, medically supervised medications to reduce withdrawal symptoms, prevent relapse, and decrease opioid-related harm. ORT includes both opioid agonists such as methadone and buprenorphine and opioid antagonists such as naltrexone, offering a flexible treatment approach that either stabilizes opioid dependence or supports complete abstinence.
On the other hand, opioid agonist therapy (OAT) is a subset of ORT that specifically uses opioid agonists—medications that activate opioid receptors in the brain. The goal of OAT is to relieve withdrawal symptoms and cravings without producing the intense euphoria of illicit opioids. OAT includes full agonists like methadone, which strongly activates opioid receptors, and partial agonists like buprenorphine, which activate receptors to a lesser extent, reducing the risk of overdose.


