Vaccines against addiction: do they exist, history, current research, and difficulties

Vaccines against addiction are currently unavailable as an approved treatment modality. There are vaccines under development for multiple substances of abuse, of which only nicotine and cocaine vaccines are at the Phase III clinical trial stage.
The history of addiction vaccines goes back to the 1970s when scientists started working on vaccines for opiates. In the next few years, vaccines against morphine were developed. Research on vaccines then stopped before resuming in the 1990s, spurred by an increase in addiction to stimulants. However, the most significant work on vaccines against addiction has been carried out in the last 20 years.
The current status of research on addiction vaccines is that there is a focus on developing vaccines with enhanced immunogenicity using nanoparticles, polymers as carriers, synthetic haptens, and adjuvants that are able to elicit powerful immune responses. Researchers are also working on developing multivalent vaccines.
The difficulties in developing addiction vaccines include suboptimal haptens and adjuvants, cross-reactivities triggered by haptens, and poor antibody response. The need to develop multivalent vaccines and overcome the difficulties of using protein-based carriers also proves challenging. Maintaining cost-effectiveness, ensuring patient compliance, conducting complex clinical trials, and improving vaccine efficacy are additional challenges.
Do vaccines against addiction currently exist?
No, vaccines against addiction do not exist currently as a treatment option approved by the Food and Drug Administration. There are multiple vaccines currently under development for various substances of abuse. A few have reached the phase of human clinical trials, and among these, only nicotine and cocaine vaccines have progressed to the stage of Phase III clinical trials, according to a 2022 article by Muhammet Celik and Brian Fuehrlein published in the journal ImmunoTargets and Therapy, titled “A Review of Immunotherapeutic Approaches for Substance Use Disorders: Current Status and Future Prospects.”
Pre-clinical trials of vaccines against addiction have shown promising results. On the other hand, current medication-assisted addiction treatment has its limitations, most notably, the possibility of precipitating complex withdrawal symptoms and the development of dependence. Behavioral therapies have significant dropout and relapse rates. In this context, addiction vaccines have the potential to fulfill the need for innovative and more effective interventions, according to a 2024 article by Lu et al., published in the journal Pharmaceutics, titled “Vaccines to Treat Substance Use Disorders: Current Status and Future Directions.” However, the challenges of developing and implementing these vaccines need to be overcome first.
What is the history of vaccines against addiction?

The history of vaccines against addiction goes back to the 1970s when scientists began thinking about how they could modify the molecules of substances of abuse like nicotine, heroin, cocaine, and amphetamines. The earliest work on vaccines was carried out for opiates, according to a 2011 article by Shen et al., published in the journal Clinical Pharmacology & Therapeutics, titled “Vaccines for Drug Abuse.”
In 1970, Spector and Parker developed a vaccine against morphine. Their achievement was followed by the development of a conjugate vaccine by Hill in 1975, according to a 2014 article by Berma Kinsey published in the journal Therapeutic Advances in Vaccines, titled “Vaccines against drugs of abuse: where are we now?” This vaccine was found to neutralize morphine in vaccinated rabbits.
However, no clinical trials of these vaccines were carried out because methadone, a synthetic opioid that blocks the effects of heroin, became available and began to be used extensively and successfully against heroin addiction. Work on developing vaccines against addiction was resumed during the 1990s when addiction to stimulants like cocaine and methamphetamine took on epidemic proportions. Since then, significant progress has been made in this field, and most of it in the last 20 years.
How would addiction vaccines work?
Addiction vaccines would work via a pharmacokinetic mechanism that prevents the drug of abuse from penetrating the blood-brain barrier and reaching the brain, according to a 2013 article by Thomas R. Kosten and Coreen B. Domingo published in the journal Expert Opinion on Biological Therapy, titled “Can you vaccinate against substance abuse?” These vaccines essentially act as blockers and work in a two-step process. Firstly, they stimulate the immune system to produce antibodies specific to the target drug. In the second stage, these antibodies bind to the target drug when taken in by the individual and prevent them from reaching the brain and other critical organs and binding to their receptors.
Most drugs of abuse have low molecular weight. They are minuscule and are able to permeate the blood-brain barrier easily to reach the brain. Antibodies are large-sized molecules and are unable to cross the blood-brain barrier. So, any drug that is bound to an antibody is unable to reach the brain, explains Paul R. Pentel in the section titled “Vaccines and Depot Medications for Drug Addiction: Rationale, Mechanisms of Action, and Treatment Implications” in the 2004 book published by the National Academies Press (US). By their nature, substances of abuse are unable to elicit an immune response or trigger the production of antibodies. So, haptens are designed.
Haptens are altered forms of the target drug. They are small molecules and are chemically conjugated with a carrier molecule that is capable of triggering an antibody response, as explained by Lu et al., in a 2024 article published in the journal Pharmaceutics, titled “Vaccines to Treat Substance Use Disorders: Current Status and Future Directions.” The resulting molecule comprising the target drug linked to a carrier is known as an immunogen. The immunogen is further paired with an adjuvant, a compound that enhances the antibody response.
When an individual who has a substantial amount of antibodies in their body takes a drug, a large portion of it binds to the antibody. The amount of the drug entering the brain is thus reduced. This, in turn, dampens the drug’s addictive effects, such as intoxication or euphoria. This mechanism reduces the rewarding and reinforcing effects of the drug that contribute to the development and maintenance of addiction. Drug cravings are diminished as the individual is less motivated to continue using the drug. Thus, anti-drug vaccines would be able to prevent relapses.
Unlike medications like naltrexone for alcohol dependence; opiate agonists and antagonists for opiate dependence; and nicotine replacement therapy and bupropion for nicotine dependence, addiction vaccines work by targeting the drug of abuse instead of the brain. The medications target areas of the brain that, apart from being implicated in addiction, are also associated with essential neurological processes like memory, cognition, movement, and emotions. Adverse side effects are not uncommon with these medications. Anti-drug vaccines are safe and produce minimal side effects because they do not act on the receptors in the brain or other organs. Unlike pharmacological agents, vaccines also do not produce dependence or withdrawal.
What is the current status of research on vaccines against addiction?
The current status of research on vaccines against addiction is that researchers are working to develop vaccines with improved immunogenicity using nanoparticles, synthetic haptens, and adjuvants with enhanced immunogenic properties, according to a 2022 article by Muhammet Celik and Brian Fuehrlein published in the journal ImmunoTargets and Therapy, titled “A Review of Immunotherapeutic Approaches for Substance Use Disorders: Current Status and Future Prospects.” Scientists are focusing on overcoming the challenges presented by the use of protein-based carriers by utilizing polymers for hapten conjugation, according to a 2024 article by Wang et al., published in the journal Zoological Research, titled “Next-generation vaccines for substance use disorders.” Currently, research is taking place to produce polyvalent vaccines that would be effective against multiple drugs of abuse.
What is TA-CD?

TA-CD is a vaccine for cocaine addiction. It is a conjugate vaccine that comprises succinyl norcocaine as a hapten that is linked to an inactivated cholera toxin B. The goal of the vaccine is to trigger the production of anti-cocaine antibodies. To date, it is one of the only two cocaine vaccines that has progressed to the clinical trial stage, according to a 2023 article by Luba et al., published in the journal Expert Opinion on Investigational Drugs, titled “Immunotherapeutic Strategies for Treating Opioid Use Disorder and Overdose.”
In the Phase I trial of TA-CD, all vaccinated subjects reported a decrease in cocaine-induced euphoria, which is the result of the vaccine-generated antibodies blocking the effects of the drug, according to a 2013 article by Kosten et al., published in the British Journal of Clinical Pharmacology, titled “Vaccines against stimulants: cocaine and MA.” The immune response lasted 2-4 months after the last vaccine dose was administered. A few subjects reported adverse side effects like hypertension, raised temperature, and mild tachycardia. The outpatient subjects who were immunized with high doses of the vaccine reported more instances of decreased relapse rates compared to those who received a low-dose vaccine.
A Phase IIa trial of TA-CD showed that there was a positive relationship between achieving a high concentration of anti-cocaine antibodies and decreased cocaine use, according to a 2022 article by Muhammet Celik and Brian Fuehrlein published in the journal ImmunoTargets and Therapy, titled “A Review of Immunotherapeutic Approaches for Substance Use Disorders: Current Status and Future Prospects.” However, only 38% of the subjects tested developed an adequate concentration of antibodies that provided protection for only two months. A Phase III clinical trial showed that 24% of those vaccinated remained abstinent during the two weeks of the trial compared to 18% in the placebo group. The group with the highest concentration of antibodies provided the highest number of cocaine-free urine samples.
However, these numbers are statistically insignificant. Besides, this cocaine addiction vaccine did not produce a decrease in cocaine use among outpatient subjects. It is believed that the TA-CD vaccine has limited therapeutic effectiveness due to the large variability in antibody concentrations among the subjects, according to a 2024 article by Lu et al., published in the journal Pharmaceutics, titled “Vaccines to Treat Substance Use Disorders: Current Status and Future Directions.”
Current research work on the TA-CD vaccine focuses on enhancing the efficacy of the vaccine by choosing or designing the right hapten and adjuvant.
What is TA-NIC?
TA-NIC is a nicotine vaccine that was developed in 1997 by Celtic Pharmaceutical. It is a conjugate vaccine comprising a nicotine hapten chemically linked to a recombinant cholera toxin-B subunit as the carrier protein.
The TA-NIC vaccine was found to produce abstinence in 38% of the subjects on their last visit or at 12 months compared to 8% in the placebo group in Phase I clinical trials, according to a 2022 article by Muhammet Celik and Brian Fuehrlein published in the journal ImmunoTargets and Therapy, titled “A Review of Immunotherapeutic Approaches for Substance Use Disorders: Current Status and Future Prospects.” The results of the Phase II clinical trials that were conducted in 2009 have not yet been published.
It is believed that the TA-NIC vaccine is effective against nicotine addiction, safe, and well-tolerated in patients, according to a 2022 article by Scendoni et al., published in the journal Human Vaccines & Immunotherapeutics, titled “Vaccines as a preventive tool for substance use disorder: A systematic review including a meta-analysis on nicotine vaccines’ immunogenicity.”
However, TA-NIC has been discontinued following its inability to achieve specified endpoints, according to a 2022 article by Hossain et al., published in the journal Vaccines, titled “Immunotherapies for the Treatment of Drug Addiction.”
What are some other vaccines under development for addiction treatment?
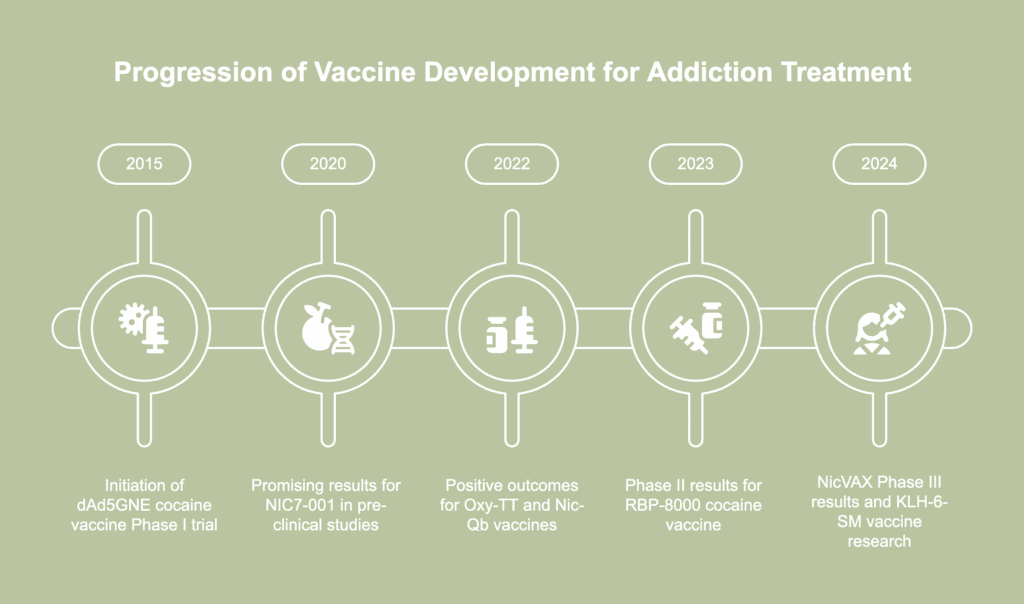
Some other vaccines under development for addiction treatment are listed below.
- Nicotine vaccines: The Phase II clinical trials of NicVAX, developed by Nabi Biopharmaceuticals, have shown that the vaccine induces the production of high levels of anti-nicotine antibodies and a substantial decrease in the occupancy of neural receptors by nicotine, according to a 2024 article by Lu et al., published in the journal Pharmaceutics, titled “Vaccines to Treat Substance Use Disorders: Current Status and Future Directions.” However, the Phase III results have not been promising, and currently, efforts are underway to reach the goal of higher abstinence with a more effective version. The Phase IIb clinical trials of the vaccine Nic-Qb (NIC002) have shown the nicotine abstinence rates in the vaccine group to be 20.2% more than that of the placebo group, according to a 2022 article by Muhammet Celik and Brian Fuehrlein published in the journal ImmunoTargets and Therapy, titled “A Review of Immunotherapeutic Approaches for Substance Use Disorders: Current Status and Future Prospects.” The vaccine NIC7-001 with a new hapten design has shown promising results in pre-clinical animal studies, producing elevated levels of nicotine-binding antibodies. It is currently being tested in a Phase I clinical study according to a 2020 review article by Schijns et al., published in the journal Immunological Reviews, titled “Modulation of immune responses using adjuvants to facilitate therapeutic vaccination.Vaccines Nic-Qb (NIC002), Nic311, and NIC7-003 have produced promising results, such as higher levels of antibodies and reduced levels of nicotine in the brain, in pre-clinical animal studies. The N4N is believed to have a higher nicotine affinity than NicVAX, but it has not yet been tested clinically. Other nicotine vaccines that have shown favorable outcomes include the nanoparticle-based NanoNicVac, which lowered brain nicotine levels in mice, and the NanoNicVac 20.0, which elicited a stronger immune response in mice and has an enhanced safety and stability profile (Lu et al., 2024). Two nicotine vaccines, PLA and PLGA, have demonstrated favorable outcomes in terms of persistence of the nicotine-binding antibodies in immunized mice for up to three months when they are administered together.
- Cocaine vaccines: A Phase I clinical trial of the vaccine dAd5GNE was initiated in 2015, with an estimated completion date in December 2025, according to a 2023 article by Luba et al., published in the journal Expert Opinion on Investigational Drugs, titled “Immunotherapeutic Strategies for Treating Opioid Use Disorder and Overdose.” A Phase II clinical trial of the vaccine TV-1380 showed a dose-dependent increase in abstinence rates in the vaccine group compared to the placebo group (Celik and Fuehrlein, 2022). The vaccine RBP-8000 demonstrated up to a 90% reduction in the plasma concentration of cocaine and physiological manifestations of the drug in a Phase II clinical trial. Pre-clinical animal studies on the vaccine GNCgzk IgG showed that it was able to prevent cocaine from triggering acute toxic and lethal effects in mice. The vaccine AAVrh.10 antiCoc.Mab was shown to sequester cocaine in blood and reduce the manifestation of unpleasant behavioral symptoms in mice.
- Heroin/morphine vaccines: Heroin tends to metabolize to three compounds, which are morphine, 6-mono-acetylmorphine [6-MAM], and morphine-6-glucuronide. The need was felt for a vaccine for heroin addiction that would be effective on multiple compounds–a multivalent vaccine. The M-KLH vaccine was found to be effective in reducing concentrations of heroin, morphine, and 6-MAM in the brains of vaccinated rodents (Luba et al., 2023). M-KLH reduced heroin-induced locomotor activity and prevented heroin-influenced reinstatement. The vaccine Her–KLH was found to block drug activity and prevent drug abuse in vaccinated rats. The Heroin-HIV-1 (H2) vaccine produced high antibody responses and decreased locomotor activity in immunized animals. The M(Gly)4-KLH vaccine was shown to reduce heroin-triggered locomotor activity in the brains of experimental rodents. The KLH-6-SM (keyhole limpet hemocyanin-6-succinylmorphine) vaccine was found to produce morphine antibodies, reduce the behavioral symptoms of the drug, and decrease the brain concentration of morphine by 25% in vaccinated rats (Celik and Fuehrlein, 2022). Currently, research is underway on the KLH-6-SM vaccine to determine if it has the potential to be used against opioid dependence depending on which heroin metabolite it shows affinity for (Lu et al., 2024).
- Oxycodone/hydrocodone vaccines: The (6OXY(Gly)4–KLH) vaccine has shown positive outcomes like increased drug binding and reduced drug distribution for both oxycodone and hydrocodone in animal studies. The OXY-dKLH vaccine effectively corrected oxycodone-triggered heart rate hold and respiratory depression in immunized animals (Lu et al., 2024). In pre-clinical animal studies on the oxycodone vaccine Oxy-TT, the vaccine group had a 50% lower brain concentration of the drug and decreased self-administration compared to the placebo group (Celik and Fuehrlein, 2022). The oxycodone vaccine (Oxy(Gly)4-sKLH) is undergoing its clinical trial phase, the only opioid vaccine to reach this stage. The results of the trials are awaited. The vaccines currently being developed and tested for oxycodone and hydrocodone produce antibodies that do not bind to off-target opioids, are not affected by opioid antagonists, and do not hinder the action of naloxone in managing an overdose (Luba et al., 2023).
- Fentanyl vaccines: It was demonstrated during pre-clinical studies on mice that the fentanyl vaccine FEN-CRM has the ability to reduce the amount of drug that enters the central nervous system. In similar animal experiments, the Fentanyl-TT vaccine produced significant dampening of fentanyl reinforcement and offered substantial protection from fatal fentanyl overdoses in vaccinated rodents (Celik and Fuehrlein, 2022). In another pre-clinical animal study, the FEN-CRM + dmLT vaccine produced large amounts of anti-fentanyl antibodies (Lu et al., 2024). These antibodies exhibited binding affinity to fentanyl and sufentanil but did not interact with morphine, methadone, oxycodone, or buprenorphine. In another study on mice, the vaccines Carfen-ester-TT and Carfen-p-phenyl-TT produced antibodies with high binding affinity to fentanyl and carfentanil. The focus of current research on fentanyl vaccines is on the development of a vaccine that has high efficacy against fentanyl and fentanyl analogs.
- Methamphetamine vaccines: The results of clinical trials of the vaccine Anti-METH mAb7F9 (ch-mAb7F9(IXT-m200)) have not yet been published. However, several vaccines for methamphetamine addiction have shown promising results in pre-clinical animal trials (Celik and Fuehrlein, 2022). The MH6-KLH vaccine was found to produce positive outcomes related to plasma concentrations of the drug and antibodies in rats. The vaccine produced a reduction in drug-triggered physiological symptoms and lowered the concentration of the drug in the brains of the animals. The SMA-TT vaccine lowered drug concentrations in mice and reduced acquisition and methamphetamine self-administration.
What are the difficulties in developing vaccines for addiction?
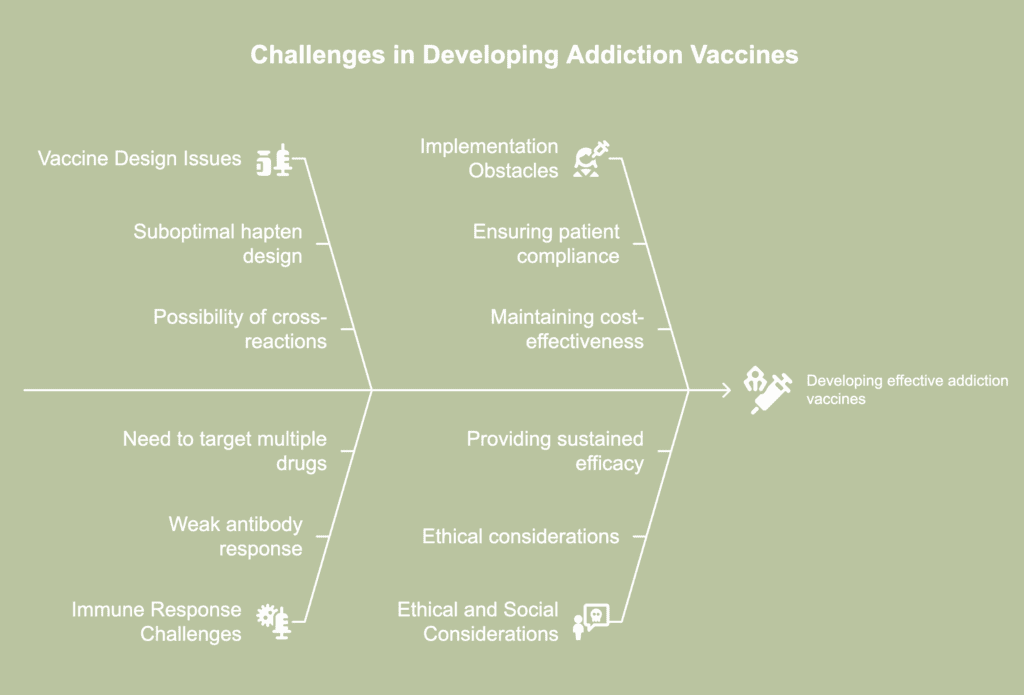
The difficulties in developing vaccines for addiction are listed below.
- Suboptimal hapten design: Haptens are modified forms of the addictive substance against which a vaccine needs to be created. When conjugated with a carrier, these are able to produce an immune reaction when introduced into the body. The challenge is to produce a hapten that retains the essential structural features of the target drug, according to a 2024 article by Lu et al., published in the journal Pharmaceutics, titled “Vaccines to Treat Substance Use Disorders: Current Status and Future Directions.”
- Possibility of cross-reactions: It is possible that poorly designed haptens turn out to be structurally similar to endogenous molecules, or naturally occurring molecules within the body. These haptens then trigger cross-reactions with the natural cellular components of the subject (Lu et al., 2024).
- Drawbacks of protein-based carriers: Haptens have to be chemically bonded with a carrier molecule, generally a protein, that enables them to be recognized quickly by the immune system and thereby, elicit an antibody response. However, protein-based carriers have a few disadvantages, according to a 2024 article by Wang et al., published in the journal Zoological Research, titled “Next-generation vaccines for substance use disorders.” They cannot be adjusted physically or chemically to support personalized drug delivery mechanisms. The same drawback also makes them unsuitable for use in the development of vaccines for diverse types of addictive substances. Moreover, they carry the risk of biological contamination, degrade quickly, and have little stability. Producing and manufacturing protein-based carriers is complex. These drawbacks hinder the development of an addiction vaccine that is highly efficient, stable, and safe.
- Poor adjuvant selection: Adjuvants are compounds that are blended with the vaccine to elicit a stronger immune response from the body. It is critical that the researcher uses an adjuvant that optimizes the effectiveness of the vaccine (Lu et al., 2024). The challenge is to create an adjuvant that balances efficiency with tissue toxicity. Adjuvants that trigger the production of higher concentrations of antibodies also induce greater tissue irritation that manifests as, for example, abscesses forming at the injection site.
- Weak antibody response: It has been found that there are significant variations in the way an individual’s immune system responds to the administration of addiction vaccines. For instance, specific populations of vaccinated cocaine users do not produce adequate concentrations of antibodies that substantially dampens the effects of cocaine, according to a 2012 article by Young et al., published in the journal Nature Immunology, titled “Immune to addiction: the ethical dimensions of vaccines against substance abuse.” It is also possible that vaccines elicit weak immune responses in individuals with active ailments like hepatitis, HIV infections, and AIDS, which tend to be common among those who inject drugs. Weak antibody response fails to reduce drug cravings significantly. A section of individuals will continue to use the substance at progressively higher doses, thereby aggravating their addiction issues.
- Requirement to target multiple drugs: Opioid users tend to switch between different opioids when their specific drug of choice is not available to them. So, multivalent vaccines need to be developed that work against multiple types of opioids (Lu et al., 2024). The challenge is to develop a vaccine that produces antibodies that bind to both the parent drug compound and its active metabolites.
- Need to maintain cost-effectiveness: Young et al., (2012) note that a specific formulation of a cocaine vaccine was found to produce significant concentrations of antibody after two weeks into treatment. Furthermore, the serum concentration of antibodies could be maintained at functional levels only with regular administration of the vaccine. This reduces the cost-effectiveness of the vaccine and makes the prospect of administering the vaccine as a preventive measure less feasible.
- Ensuring patient compliance: The concentration of antibodies tends to diminish over time unless increased periodically with booster doses of the vaccine. Although there is no specific frequency of boosting, it is believed that booster doses need to be delivered every 2-6 months, as noted in the section titled “Vaccines and Depot Medications for Drug Addiction: Rationale, Mechanisms of Action, and Treatment Implications” written by Paul R. Pentel and featured in the 2004 book published by the National Academies Press (US).The administration of booster doses, which contributes to vaccine efficacy, is dependent upon ensuring patient compliance with the therapy.
- Complexity of designing clinical trials: Designingclinical trials for anti-drug vaccines is complex because of the need to accommodate the medical needs of the participants who are dependent on chemicals, according to a 2019 article by Marco Pravetoni and Sandra D. Comer published in the journal Neuropharmacology, titled “Development of vaccines to treat opioid use disorders and reduce incidence of overdose.” The most appropriate maintenance medication has to be chosen to prevent withdrawal. The challenge is to design a maintenance therapy where the medication does not cross-react with the antibodies produced by the vaccine. Designing clinical trials for anti-drug vaccines is also challenging because multiple booster doses are required to maintain optimum antibody concentrations.
- Maintaining effectiveness in active users: It is believed that vaccine efficacy is reduced in individuals who are active users of the target drug. The efficacy of anti-drug vaccines depends on their ability to rapidly and selectively bind with the target drug. Active drug users are continuously flooding their bodies with the substance, in amounts far greater than what the available antibodies are able to bind to. Hence, the efficacy of the vaccine is reduced.
- P roviding sustained efficacy: Drug abuse is only partly the result of biological factors. It is more an outcome of multiple socio-economic and cultural factors and stigma and discrimination at individual, social, and institutional levels (Young et al., 2012). Achieving long-term abstinence depends on addressing the environmental and behavioral aspects of addiction, which a vaccine cannot achieve on its own.
- Ethical considerations: It is possible that an anti-drug vaccine is perceived as a “magic bullet” that resolves the problem of addiction. It is critical to clarify and educate that getting vaccinated is not a license to continue abusing drugs. Vaccines do not eliminate the need to address the social, behavioral, and environmental components of a multifactorial disease like addiction.


